Hyaluronidases and Dissolving Filler
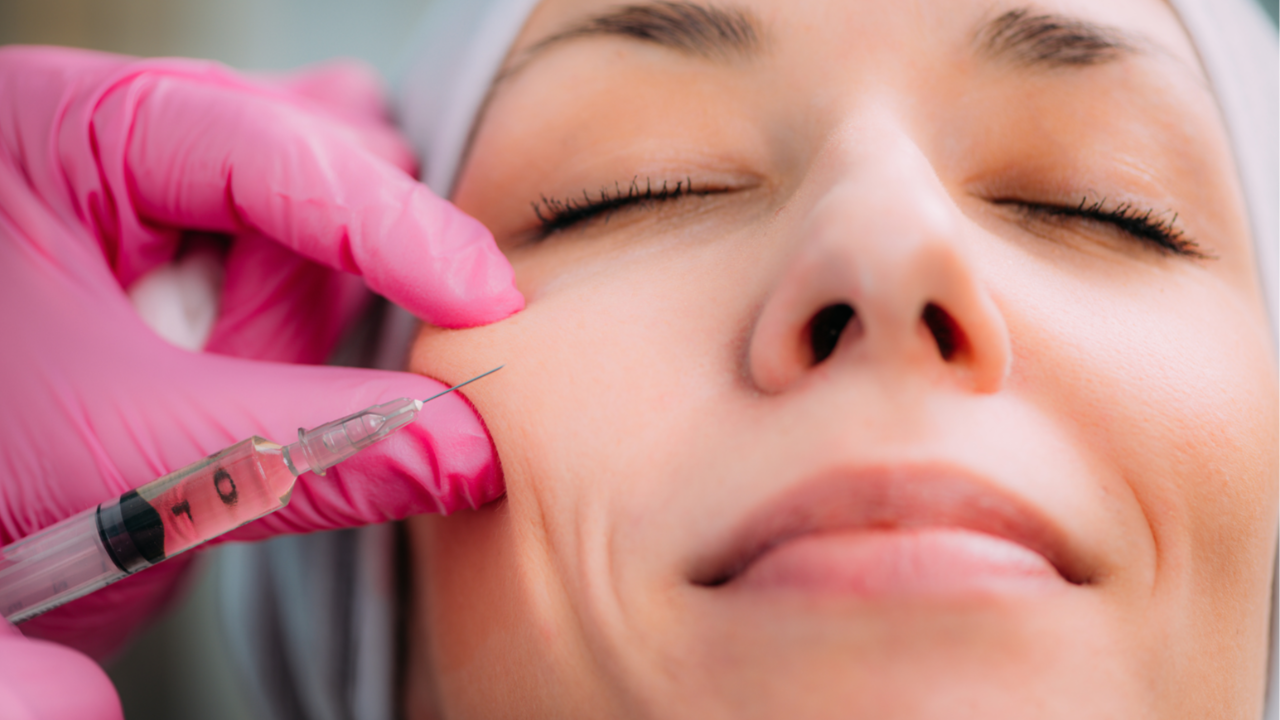
Natural looks are trending, and as more people open up about getting injectables, many are reversing the effects of their fillers. While injections may fade over time, hyaluronidases can help speed up the process.
What are hyaluronidases?
Hyaluronidases are a family of enzymes that break down hyaluronic acid (HA). The body degrades HA via thermal pathways, free radicals, and naturally occurring hyaluronidases. While these natural processes will help dissolve the filler over time, patients finding complications with their treatment may look for alternative methods of dissolution.
When to get hyaluronidase injections
Adverse effects and complications from hyaluronic acid fillers may include the following:
- Cosmetic overcorrection
- Inflammatory nodules
- Delayed onset nodules
- Vascular occlusion
- Blindness
- Tyndall effect
- Allergic or immunogenic reaction to HA filler
- Necrosis
Each of these occurrences may prompt patients to seek correction via hyaluronidase. Unless the patient is experiencing necrosis or another severe condition which should be treated within 4 hours, patients must wait at least a day after receiving a filler injection to receive hyaluronidase correction. Times may vary based on injector recommendation.
To dissolve HA fillers, concentrated synthetic hyaluronidases are injected back into the filler sites for reversal. The effects are almost immediately noticeable, as the hyaluronidase will get to work right away. Patients will see the effects continue for 24 to 48 hours, before symptoms subside. Upon injector recommendation, patients may wait up to two weeks at most before adding more filler post hyaluronidase treatment. Timelines will vary based on degree of swelling and bruising.
Types of hyaluronidases
HA is found in some of the most popular filler brands like Restylane and Juvederm. Restylane was found to be more sensitive than Juvederm to multiple types of hyaluronidases.
Hyaluronidases are classified into two types based on their activation in varying pH levels. Acid-active hyaluronidases are activated at a pH of 3 to 4. Neutral-active hyaluronidases are activated in a pH of 5 to 8.
There are four main brands of hyaluronidases used to treat dermal fillers:
- Hylenex: synthetic, recombinant DNA made from human products
- Hydase: made from cows
- Amphadase: made from cows
- Vitrase: made from sheep products
How much hyaluronidase to use
Units of hyaluronidase will vary based on the amount and type of filler the patient received as well as the region. As indicated by JCAD Aesthetic Complications guidelines by the ACE Group, the following formula may be used as a general guideline:
Volume to inject (mLs)=Number of units required (units)Total number of units (1500 units)Volume of diluent (mL)
A general consensus determined by a comprehensive review from the ACE Group states that five units of hyaluronidase is needed to break down 0.1mL of 20mg/mL HA. However, recommendations vary by case.
To treat vascular occlusion or other ocular indications, units are recommended by region as follows:
|
Region |
Hyaluronidase (Units) |
|
Nasal and perioral skin |
15-30 |
|
Periorbital |
3-4.5 |
|
Infraorbital |
10-15 |
|
Lower lid |
1.5 |
How to inject hyaluronidase
An injector should inspect, palpate, and mark out the area they intend to inject. It should then be cleansed and disinfected before injection with a 27G or 30G needle of appropriate length by area of the body. Because the enzyme cannot distinguish between naturally occurring HA in the skin and artificially injected HA from the filler, doses are administered only at the filler injection sites.
No local anesthetic should be injected, as it may result in complications. When lidocaine was used in combination with hyaluronidase, patients experienced a significant increase in trismus (lockjaw) and postoperative pain.
Post treatment side effects
Post hyaluronidase treatment, patients might see some slight bruising and swelling. This can be mitigated by massaging the injection site and applying heat, which will also promote degradation of the filler. All symptoms should resolve within 24 to 48 hours.
Neutral-active hyaluronidases are present in snake and bee venom, prompting extensive testing on allergic reactions as a result of dissolving filler. However, rates of allergic reactions are extremely rare at a frequency of 0.05% to 0.69%.
Non-hyaluronic acid fillers cannot be dissolved by hyaluronidases. Lumpiness or asymmetry may be corrected by injecting more filler to fix the shape. Other options include surgical removal or allowing the fillers to fade on their own.
Works Referenced
- King, Martyn, et al. “The Use of Hyaluronidase in Aesthetic Practice (v2.4).” JCAD The Journal of Clinical and Aesthetic Dermatology, 1 June 2018, jcadonline.com/aesthetic-complications-hyaluronidase/.
- L;, Cavallini M;Gazzola R;Metalla M;Vaienti. “The Role of Hyaluronidase in the Treatment of Complications from Hyaluronic Acid Dermal Fillers.” Aesthetic Surgery Journal, pubmed.ncbi.nlm.nih.gov/24197934/.
- M;, Landau. “Hyaluronidase Caveats in Treating Filler Complications.” Dermatologic Surgery : Official Publication for American Society for Dermatologic Surgery [et Al.], pubmed.ncbi.nlm.nih.gov/26618463/.
- Ridenour, S, et al. “Anesthetic Efficacy of a Combination of Hyaluronidase and Lidocaine with Epinephrine in Inferior Alveolar Nerve Blocks.” Anesthesia Progress, 2001, www.ncbi.nlm.nih.gov/pmc/articles/PMC2007333/.


